 
Two spheres are pushed very tightly together. The first model, in light green, is used to find the F atom radius. In figure a, 4 diatomic molecules are shown to illustrate the method of determining the atomic radius of an atom. The general trend is that radii increase down a group and decrease across a period. (b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. Therefore, the valence electrons of krypton are eight.\): (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. 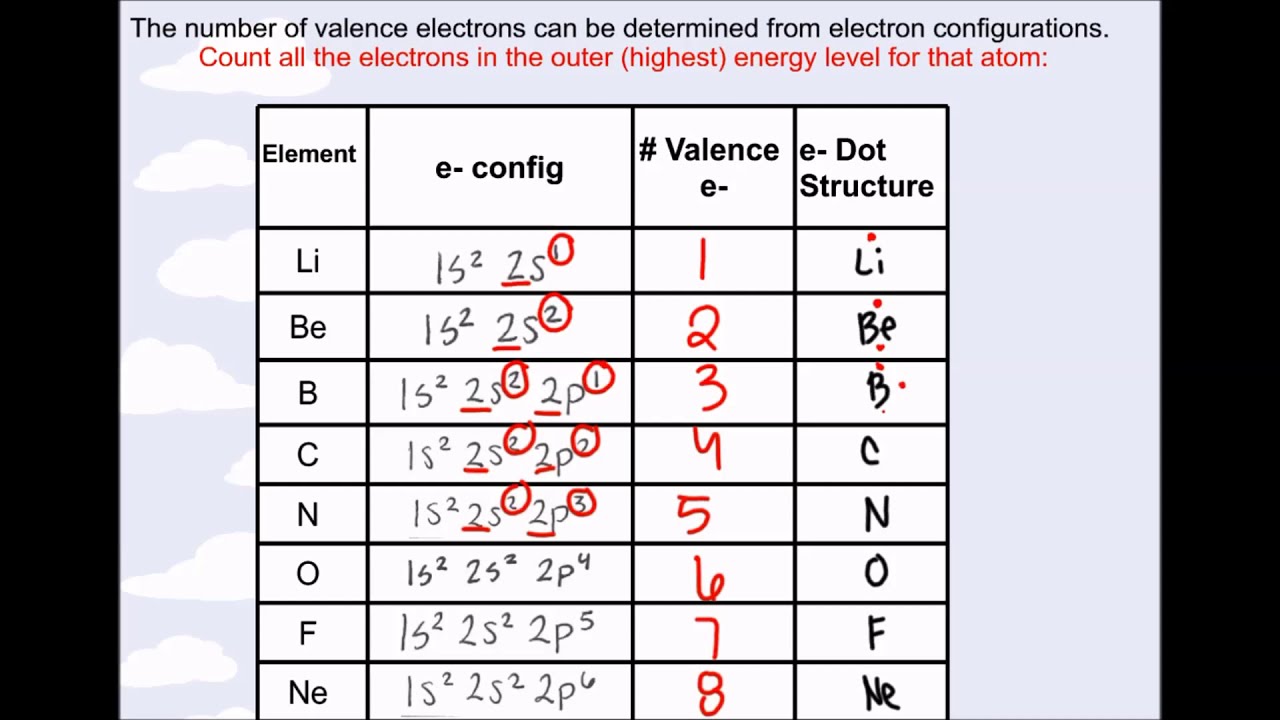
The electron configuration shows that the last shell of krypton has eight electrons. The total number of electrons in a valence shell is called valence electrons. The last shell after the electron configuration is called the valence shell. The third step is to diagnose the valence shell. Step-3: Determine the valence shell and calculate the total electrons The number of electrons per shell of krypton is 2, 8, 18, 8. That is, the first shell of krypton has two electrons, the second shell has eight electrons, the 3rd shell has eighteen electrons and the 4th shell has eight electrons. The electron configuration of the krypton shows that there are two electrons in the K shell, eight in the L shell, eighteen in the M shell, and eight in the N shell. We know that krypton atoms have a total of thirty-six electrons. In this step, the electrons of krypton have to be arranged. Step-2: Need to do electron configuration of krypton That is, the krypton atom has a total of thirty-six electrons. From the periodic table, we see that the atomic number of krypton is 36. That is, we can finally say that there are electrons equal to the atomic number in the krypton(Kr) atom. Position of krypton(Kr) in the periodic table And electrons equal to protons are located outside the nucleus. The atomic number is the number of protons. It is necessary to know the atomic number of krypton elements from the periodic table. To know the atomic number we need to take the help of a periodic table. To know the number of electrons, you need to know the number of protons in krypton.Īnd to know the number of protons, you need to know the atomic number of the krypton element. Step-1: Determining the total number of electrons in kryptonġst we need to know the total number of electrons in the krypton atom. Now we will learn how to determine the valence electron of krypton. However, valence electrons can be easily identified by arranging electrons according to the Bohr principle. Knowing the electron configuration in the right way, it is very easy to determine the valence electrons of all the elements. 
It is not possible to determine the valence electron without electron configuration. The electron configuration is one of them. The valence electrons have to be determined by following a few steps. How do you calculate the number of valence electrons in a krypton atom? The valence electrons determine the properties of the element and participate in the formation of bonds. The total number of electrons in the last shell after the electron configuration of krypton is called the valence electrons of krypton. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
